Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 -implanted GaN film studied by a monoenergetic positron beam
-implanted GaN film studied by a monoenergetic positron beamLi, X. F.*; Chen, Z. Q.*; Liu, C.*; Zhang, H.; Kawasuso, Atsuo
Journal of Applied Physics, 117(8), p.085706_1 - 085706_6, 2015/02
Times Cited Count:23 Percentile:67.91(Physics, Applied)Vacancy-type defects in C -implanted GaN were probed using a slow positron beam. The increase of Doppler broadening S parameter indicates introduction of arge vacancy clusters. Post-implantation annealing at temperatures up to 800
-implanted GaN were probed using a slow positron beam. The increase of Doppler broadening S parameter indicates introduction of arge vacancy clusters. Post-implantation annealing at temperatures up to 800 C makes these vacancy clusters to agglomerate into microvoids. The vacancy clusters or microvoids show high thermal stability, and they are only partially removed after annealing up to 1000
C makes these vacancy clusters to agglomerate into microvoids. The vacancy clusters or microvoids show high thermal stability, and they are only partially removed after annealing up to 1000 C. Amorphous regions are observed by high resolution transmission electron microscopy measurement, which directly confirms that amorphization is induced by C
C. Amorphous regions are observed by high resolution transmission electron microscopy measurement, which directly confirms that amorphization is induced by C -implantation. The disordered GaN lattice is possibly due to special feature of carbon impurities, which enhance the damage buildup during implantation.
-implantation. The disordered GaN lattice is possibly due to special feature of carbon impurities, which enhance the damage buildup during implantation.
Aihara, Jun; Hojo, Kiichi; Furuno, Shigemi*; Shimura, Kenichiro; Hojo, Tomohiro*; Sawa, Kazuhiro; Yamamoto, Hiroyuki; Motohashi, Yoshinobu*
Nuclear Instruments and Methods in Physics Research B, 242(1-2), p.441 - 444, 2006/01
Times Cited Count:1 Percentile:12.84(Instruments & Instrumentation)no abstracts in English
Aihara, Jun; Hojo, Tomohiro*; Furuno, Shigemi*; Ishihara, Masahiro; Sawa, Kazuhiro; Yamamoto, Hiroyuki; Hojo, Kiichi
Nuclear Instruments and Methods in Physics Research B, 241(1-4), p.559 - 562, 2005/12
Times Cited Count:5 Percentile:41.01(Instruments & Instrumentation)Silicon carbide (SiC) specimens prepared for the TEM (transmission electron microscope) observation were amorphized with 30keV Ne or 4.5keV He ion irradiation at room temperature and successively annealed at 1273K in the TEM. Ne and He were selected as irradiation ion species to change the concentration of implanted rare gas atoms. The energy and flux of these ion species were selected in order to get similar dpa depth profiles and dpa rates based on TRIM calculation. In this condition, peak ion implantation of He was estimated to be about 5 times as large as that of Ne for the same peak dpa. Crystal nucleation occurred with annealing in the specimen irradiated with He up to 6.3dpa(peak), however, no crystal nucleation was observed in the specimen irradiated with Ne up to 15dpa(peak); Namely, crystal nucleation occurred with less dpa in the case of He irradiation than in the case of Ne irradiation. It was found that the concentration of implanted inert gas atom influences the crystal nucleation behavior.
Aihara, Jun; Ishihara, Masahiro; Hojo, Kiichi; Furuno, Shigemi*
Journal of the American Ceramic Society, 87(6), p.1146 - 1148, 2004/06
Times Cited Count:1 Percentile:18.37(Materials Science, Ceramics)SiC specimens were amorphized with Ne irradiation and annealed at 1273K. One specimen was annealed continuously for 60 minutes, the other was annealed repeatedly (5 minutes  10 times). Crystal nucleation in the amorphized SiC was apt to occur more in the case of repeated annealing than in the case of continuous annealing.
10 times). Crystal nucleation in the amorphized SiC was apt to occur more in the case of repeated annealing than in the case of continuous annealing.
Aihara, Jun; Hojo, Kiichi; Furuno, Shigemi*; Ishihara, Masahiro; Yamamoto, Hiroyuki
Proceedings of 8th Asia-Pacific Conference on Electron Microscopy (8APEM) (CD-ROM), p.722 - 723, 2004/06
SiC TEM specimens were irradiated with 20keV Ne+ to the fluence of 1.5x10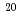 Ne+/m
Ne+/m at 573, 583, 598 and 683K, and successively annealed at 1273K for 30minutes. In the cases of 573 and 583K irradiation, amorphization with irradiation and crystal nucleation with annealing occurred. Coalescence of the bubbles was clearly observed in the crystal nucleated area and epitaxial growth area. In the case of 593K irradiation, partial amorphization occurred but crystal nucleation did not occur. In the case of 673K irradiation, amorphization did not occur and no change was observed after annealing.
at 573, 583, 598 and 683K, and successively annealed at 1273K for 30minutes. In the cases of 573 and 583K irradiation, amorphization with irradiation and crystal nucleation with annealing occurred. Coalescence of the bubbles was clearly observed in the crystal nucleated area and epitaxial growth area. In the case of 593K irradiation, partial amorphization occurred but crystal nucleation did not occur. In the case of 673K irradiation, amorphization did not occur and no change was observed after annealing.
Hojo, Tomohiro; Aihara, Jun; Hojo, Kiichi; Furuno, Shigemi*; Yamamoto, Hiroyuki; Nitani, Noriko; Yamashita, Toshiyuki; Minato, Kazuo; Sakuma, Takaaki*
Journal of Nuclear Materials, 319, p.81 - 86, 2003/06
Times Cited Count:18 Percentile:74.33(Materials Science, Multidisciplinary)no abstracts in English
 O
O and MgO irradiated with energetic iodine ions
and MgO irradiated with energetic iodine ionsAruga, Takeo; Katano, Yoshio*; Omichi, Toshihiko*; Okayasu, Satoru; Kazumata, Yukio*; Jitsukawa, Shiro
Nuclear Instruments and Methods in Physics Research B, 197(1-2), p.94 - 100, 2002/11
Times Cited Count:9 Percentile:51.38(Instruments & Instrumentation)no abstracts in English
 O
O , MgAl
, MgAl O
O and MgO irradiated with energetic iodine ions
and MgO irradiated with energetic iodine ionsAruga, Takeo; Katano, Yoshio*; Omichi, Toshihiko*; Jitsukawa, Shiro
Surface & Coatings Technology, 158-159, p.444 - 448, 2002/09
no abstracts in English
Aihara, Jun; Hojo, Kiichi; Furuno, Shigemi*; Ishihara, Masahiro; Hayashi, Kimio
Journal of Electron Microscopy, 51(2), p.93 - 98, 2002/05
Times Cited Count:6 Percentile:20.46(Microscopy)no abstracts in English
Takahiro, Katsumi*; Takeshima, Naoki*; Kawatsura, Kiyoshi*; Nagata, Shinji*; Yamamoto, Shunya; Narumi, Kazumasa; Naramoto, Hiroshi
JAERI-Review 2001-039, TIARA Annual Report 2000, p.188 - 189, 2001/11
no abstracts in English
Muramatsu, Yasuji; Hirono, Shigeru*; Umemura, Shigeru*; Ueno, Yuko*; Hayashi, Takayoshi*; Grush, M. M.*; Gullikson, E. M.*; Perera, R. C. C.*
Carbon, 39(9), p.1403 - 1407, 2001/06
Times Cited Count:18 Percentile:58.27(Chemistry, Physical)no abstracts in English
Abe, Hiroaki
Tanso Sogenryo Kagaku To Zairyo Sekkei, 3, p.5 - 14, 2001/00
no abstracts in English
Aruga, Takeo; Katano, Yoshio; Omichi, Toshihiko; Okayasu, Satoru; Kazumata, Yukio
Nuclear Instruments and Methods in Physics Research B, 166-167, p.913 - 919, 2000/05
Times Cited Count:32 Percentile:86.89(Instruments & Instrumentation)no abstracts in English
Oda, Chie; *
JNC TN8400 98-001, 14 Pages, 1998/11
Solubilities of amorphous stannic oxide, SnO (am) in Na-ClO
(am) in Na-ClO -Cl-SO
-Cl-SO aqueous systems were measured to quantitatively investigate the influences of the ligands OH
aqueous systems were measured to quantitatively investigate the influences of the ligands OH , Cl
, Cl and SO
and SO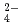 on solubilities. They were also measured in bentonite equilibrated solutions to discuss the behavior of tin under a repository condition of a high-revel radioactive waste. The solubility data in sodium perchlorate media in the range of pH from 6 to 11 showed pH dependency, and the hydrolysis constants of tin (IV) were determined (Amaya, et al., 1997). No significant changes in solubilities with the variation in Cl
on solubilities. They were also measured in bentonite equilibrated solutions to discuss the behavior of tin under a repository condition of a high-revel radioactive waste. The solubility data in sodium perchlorate media in the range of pH from 6 to 11 showed pH dependency, and the hydrolysis constants of tin (IV) were determined (Amaya, et al., 1997). No significant changes in solubilities with the variation in Cl , SO
, SO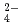 concentrations were observed in Na-ClO
concentrations were observed in Na-ClO -Cl-SO
-Cl-SO aqueous systems, so this indicates that chloride and sulfate species were less effective than hydroxide complexes. On the other hand, solubilities in bentonite equilibrated solutions were higher than solubilities of other experiments in simple systems. These results suggest that (1) other complexes of tin except hydroxide, chloride or sulfate complexes of tin (IV) may dominantly exist in aqueous phase, (2)solid phase other than SnO
aqueous systems, so this indicates that chloride and sulfate species were less effective than hydroxide complexes. On the other hand, solubilities in bentonite equilibrated solutions were higher than solubilities of other experiments in simple systems. These results suggest that (1) other complexes of tin except hydroxide, chloride or sulfate complexes of tin (IV) may dominantly exist in aqueous phase, (2)solid phase other than SnO (am) may limit the solubility of tin under repository conditions.
(am) may limit the solubility of tin under repository conditions.
 Si
SiUgajin, Mitsuhiro; Akabori, Mitsuo; Ito, Akinori; Ooka, Norikazu;
Journal of Nuclear Materials, 248, p.204 - 208, 1997/00
Times Cited Count:8 Percentile:56.29(Materials Science, Multidisciplinary)no abstracts in English
Katayama, Yoshinori
Koatsuryoku No Kagaku To Gijutsu, 6(4), p.216 - 221, 1997/00
no abstracts in English
Hojo, Kiichi; Furuno, Shigemi; *; *; *
Nuclear Instruments and Methods in Physics Research, 127-128, p.203 - 207, 1997/00
no abstracts in English
Hojo, Kiichi; *; Furuno, Shigemi; Sasajima, Naohiko*; *
Journal of Nuclear Materials, 239(1-3), p.279 - 283, 1996/00
Times Cited Count:14 Percentile:74.12(Materials Science, Multidisciplinary)no abstracts in English